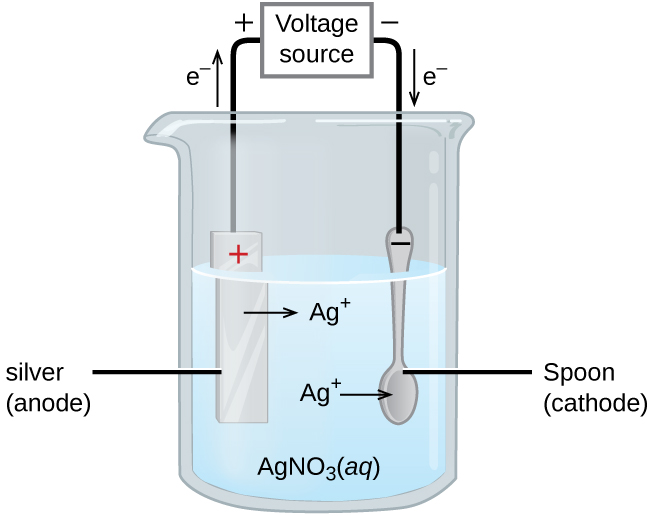
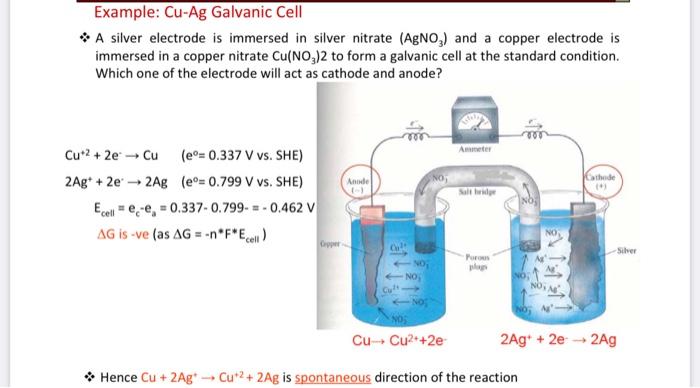
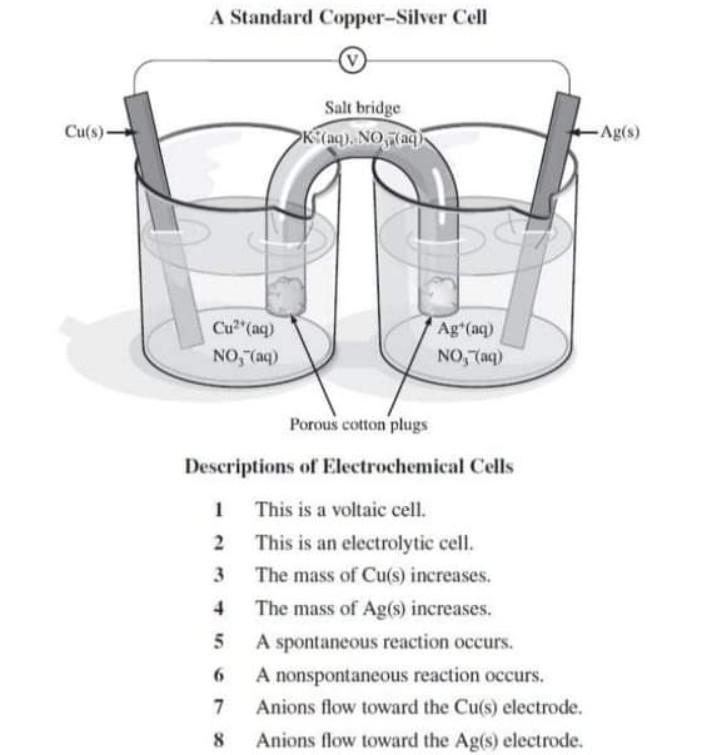
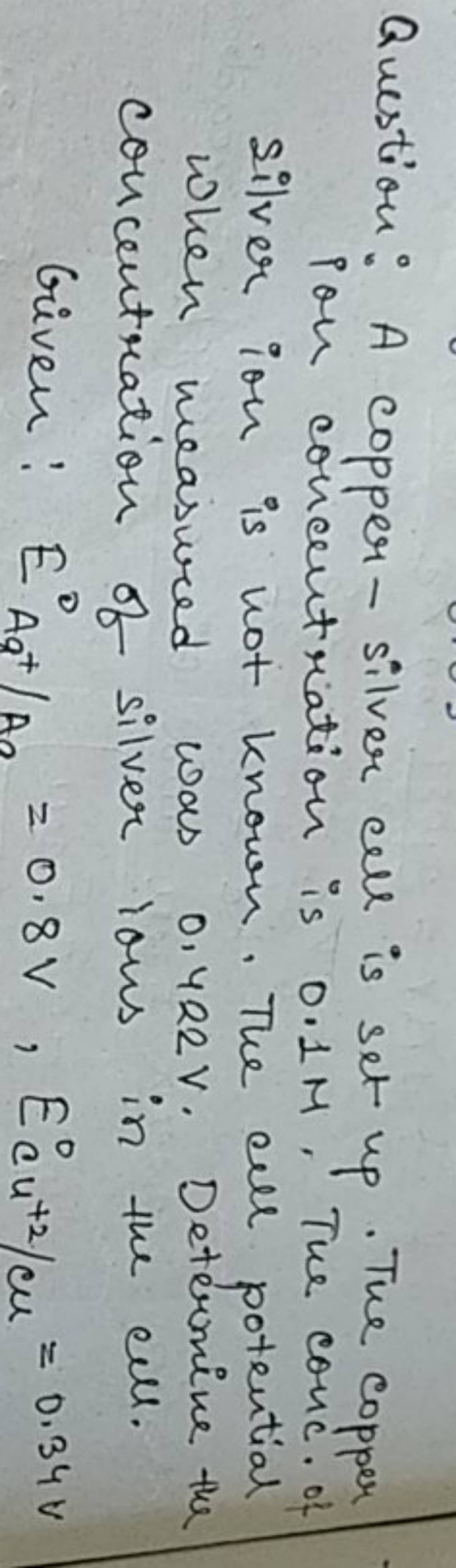7. of A copper silver cell is setup. The copper ion concentration in it is 0.10 M. The concentration of silver ion is not known. The cell potential measures 0.422V. Determine the
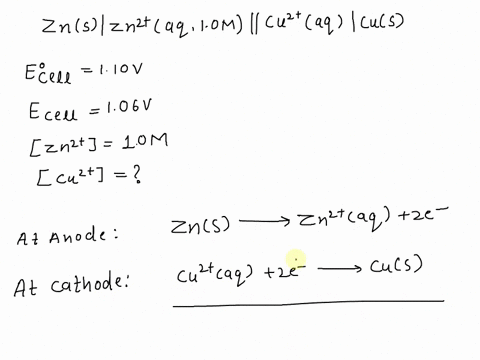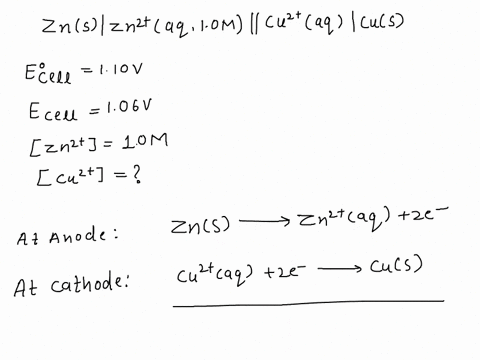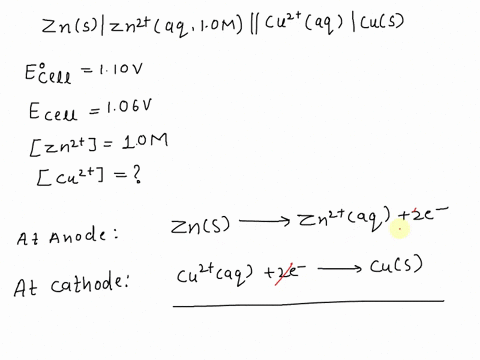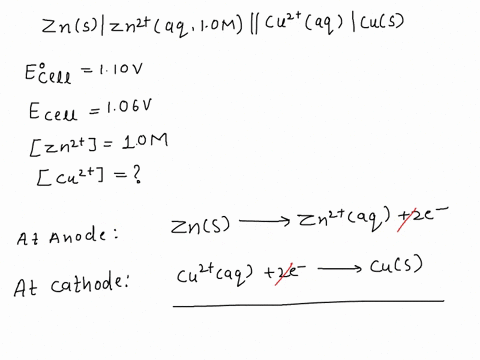
SOLVED: A copper-silver cell is set up. The copper ion concentration in it is 0.10 M. The concentration of silver ion is not known. The cell potential is measured as 0.422 V.

A copper - silver cell is set up. The copper ion concentrations is 0.10 M. The concentration of... - YouTube
A copper- silver cell is set up. The copper ion concentration in it is 0.10 M. The concentration of silver ion is not known. The cell potential measured 0.422 V. Determine the
BIM objects - Free download! Copper-Silver Ionization System - CSI - 2 Flow Cell 2 Controller Rack Mount - 2F2C | BIMobject

A copper-silver cell is set up. The copper ion concentration in it is 0.10 M. The concentration of silver - Brainly.in

SOLVED: The Cu2+ ion concentration in a copper-silver electrochemical cell is 0.5 M. If Eo (Ag+/Ag) = 0.9 V, Eo (Cu2+/Cu) = 0.34 V, and cell potential (at 25°C) = 0.422 V,

SOLVED: A copper-silver cell is set up. The copper ion concentration in it is 0.10 M. The concentration of silver ion is not known. The cell potential is measured as 0.422 V.

SOLVED: The Cu2+ ion concentration in a copper-silver electrochemical cell is 0.5 M. If Eo (Ag+/Ag) = 0.9 V, Eo (Cu2+/Cu) = 0.34 V, and cell potential (at 25°C) = 0.422 V,

Solve this: 8 A Copper-Silver cell is set up The copper ion - Chemistry - Electrochemistry - 12485096 | Meritnation.com
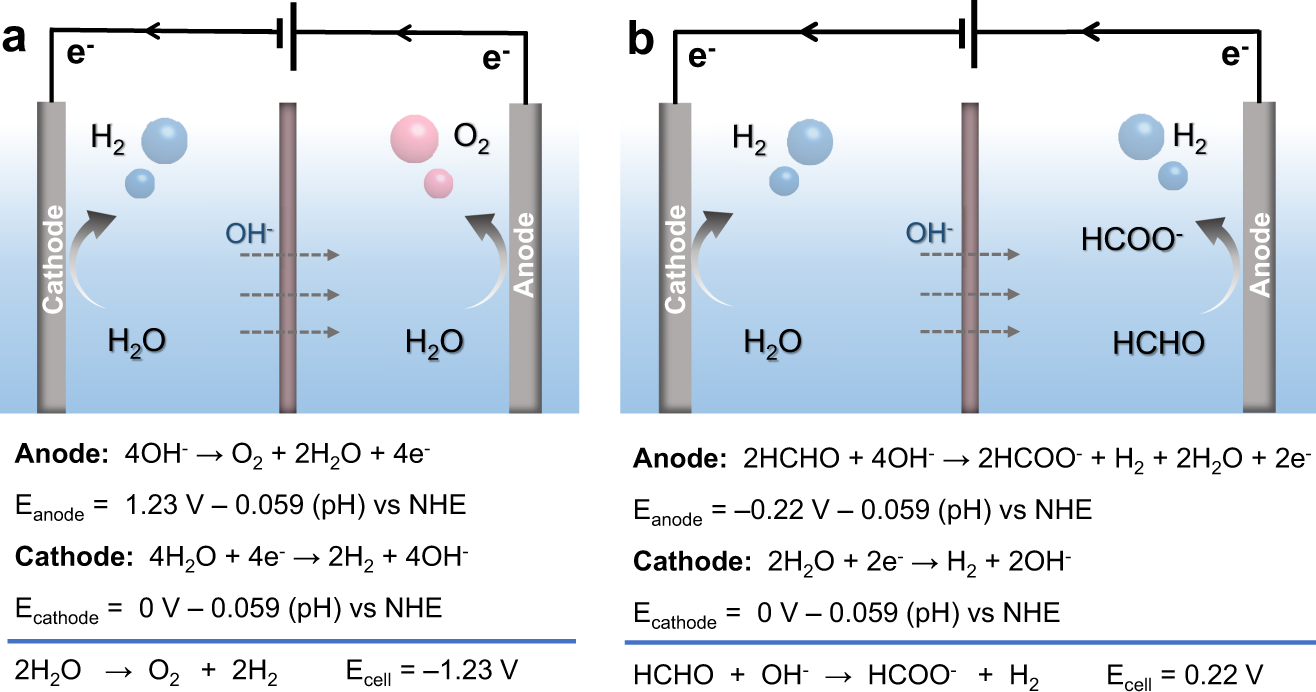
Dual hydrogen production from electrocatalytic water reduction coupled with formaldehyde oxidation via a copper-silver electrocatalyst | Nature Communications
A copper-silver cell is set up. The copper ion concentration in it is 0.10 M. The concentration of silver ion is not known. - Sarthaks eConnect | Largest Online Education Community